Early Detection

Can pancreatic cancer be detected early?
Pancreatic cancer is hard to diagnose early. There is no widespread screening for pancreatic cancer in the general population yet. However, there are imaging and blood-based tests that may be able to detect pancreatic cancer in its early stages in people who are at an increased risk for the disease or for people with symptoms who have been referred by their doctor for further tests.
Early-stage pancreatic cancer is usually found if the location of the cancer causes symptoms early or if testing for unrelated medical conditions shows signs of the disease. But most pancreatic cancer patients are diagnosed at stage IV.
Researchers across the world are working to develop early detection methods for pancreatic cancer. Those at high risk may consider research studies like surveillance programs. These programs use regular monitoring to look for the disease with the hope of finding it earlier if it does develop.

Worried about pancreatic cancer in your family?
By answering a few quick questions about your family history, we can check your family risk of developing pancreatic cancer.
Why is early detection important?
People who have pancreatic cancer diagnosed in the early stages of disease have better outcomes. If pancreatic cancer is detected before it has grown too large or spread to other areas in the body there are more treatment options available, including surgery.
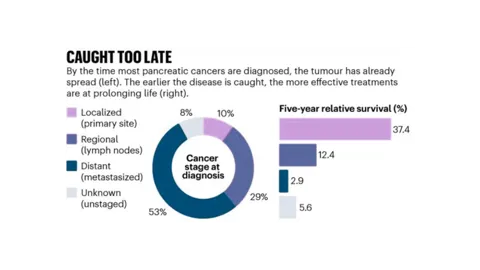
Around 10% of pancreatic cancers are diagnosed before they spread. However, if detected early, long-term (5 years +) survival rates are 10 times higher than if the cancer has already spread. It is important to remember that while survival statistics give an indication of prognosis on a population level scale, they cannot describe individual cases.
Early detection also provides greater time and opportunity for patients to participate in research, accelerating discovery and treatment options.
Source: US National Cancer Institute
Why is pancreatic cancer hard to diagnose?
- The symptoms of pancreatic cancer are not always obvious, come and go and can change over time.
- The pancreas is located deep within the body, so it can be hard to see or feel a tumour from a physical exam.
- Current diagnostic tests do not always detect small, pre-cancerous or early-stage tumours.
- While there are several diagnostic tests available such as MRI and endoscopic ultrasound, people get referred to the standard tests (such as MRI and endoscopic ultrasound) once they present symptoms and it’s often too late. There is no population based surveillance to look for pancreatic cancer as there is in breast, prostate, colon or lung cancer.
- There are no proven biological markers that can be used to determine if you have pancreatic cancer.

Early Detection Research
Since our inception, we have been committed to funding impactful research that furthers the understanding of pancreatic cancer, including projects to detect the cancer earlier and provide better treatment options to improve survival.
